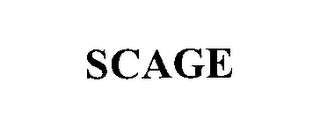Information
-
 Trademark
Trademark
-
 76346743
76346743
-
Serial Number
76346743
-
Registration Number
3081096
-
International Classifications
- 1 - Chemicals used in industry, science and photography, as well as in agriculture, horticulture and forestry
- 5 - Pharmaceutical and veterinary preparations
- 9 - Scientific, nautical, surveying, photographic, cinematographic, optical, weighing, measuring, signalling, checking (supervision), life-saving and teaching apparatus and instruments
- 10 - Surgical, medical, dental and veterinary apparatus and instruments, artificial limbs, eyes and teeth
- 42 - Scientific and technological services and research and design relating thereto
-
Filing Date
December 07, 2001
24 years ago
-
Registration Date
April 18, 2006
19 years ago
-
Transaction Date
January 01, 2013
13 years ago
-
Status Date
November 23, 2012
13 years ago
-
Published for Opposition Date
May 04, 2004
21 years ago
-
Location Date
April 18, 2006
19 years ago
-
Status Code
710
-
Current Location
PUBLICATION AND ISSUE SECTION
Employee Name
JENKINS JR, CHARLES L
-
Attorney Docket Number
DEBE:004 /SL
Attorney Name
Steven L. Highlander
Law Office Assigned Location Code
L50
-
Owners
Mark Drawing Code
1000
Mark Identification
SCAGE
Case File Statements
- GS0011: Chemical preparations, namely, buffer, polypeptides, nucleic acids for use in scientific experiments; chemical agents for characterizing and examination of diseases, particularly of tumors, coronary heart diseases, particularly restenosis, minimal residual diseases, infectious diseases, autoimmune diseases for scientific purposes; chemical agents for characterizing and examination of eukaryotic cells, namely, animal cells, particularly mammalian cells and human cells, green plant cells, fungi cells, rhodophyta cells, alveolate cells, stramenopiles cells, other protist cells, particularly kinetoplastida and amoeboflagellata, cells of microscopic small biopsies of animal and human tissues, of cells out of tissue samples prepared with a laser, of cells isolated with FACS or with receptor/ligand binding, of chemical fixed tissue, of micrometastatic cells, of bone marrow cells, of samples from body fluids, particularly from amniotic fluid, liquor, blood, lymph, urine or bowel movement, of stem cells, cloned single cells or differentiated stages from specific cells for scientific use; eukaryotic cell culture lines, namely, animal cell culture lines, particularly mammalian cell culture lines and human cell culture lines, green plant cell culture lines, fungi cell culture lines, rhodophyta cell culture lines, alveolate cell culture lines, stramenopiles cell culture lines, other protist cell culture lines, particularly kinetoplastida and amoeboflagellata cell culture lines for scientific purposes; cultures of microorganisms for scientific purposes; nucleic acids for scientific purposes
- GS0051: Medical diagnostic chemical agents for characterizing and examination of diseases, namely, tumors, coronary heart diseases, particularly restenosis, minimal residual diseases, infectious diseases, autoimmune diseases; pharmaceutical preparations for the treatment of tumors, coronary heart diseases, particularly restenosis, minimal residual diseases, infectious diseases, autoimmune diseases and veterinary preparations for the treatment of pets and farm animals, namely pigs, dogs, cats, ferrets, hoofed animals, particularly horses, cattle, sheep, asses and mules, birds, particularly poultry, particularly chickens, ducks, turkeys and gooses, pigeons, parrots and budgerigars, fishes, small animals, particularly hares, rabbits, hamsters, mice, rats and guinea pigs, reptiles, particularly snakes, frogs, turtles, crocodiles and lizards; pharmaceutical preparations for diagnosis and analysis of tumors; coronary heart diseases, particularly restenosis, minimal residual diseases, infections diseases, autoimmune diseases; pharmaceutical preparations for diagnosis and analysis of eukaryotic cells, mammalian cells and human cells, of cells of microscopic small biopsies of animal and human tissues, of cells out of tissue samples prepared with a laser, of cells isolated with FACS or with receptor/ligand binding, of cells of chemical fixed tissue, of micrometastatic cells, of bone marrow cells, of samples from body fluids, particularly amniotic fluid, from liquor, blood, lymph, urine or bowel movement, of stem cells, cloned single cells of differentiated stages from specific cells; eukaryotic cell culture lines, namely, animal cell culture lines, particularly mammalian cell culture lines and human cell culture lines, green plant cell culture lines, rhodophyta cell culture lines, alveolate cell culture lines, stramenopiles cell culture lines, protist cell culture lines, fungi cell culture lines for medical purposes; cultures of microorganisms for medical purposes; nucleic acids for medical purposes
- GS0091: Diagnostic and testing apparatus and instruments, namely, syringe, needle, biopsy needle, array, particularly DNA-array, array reader, scanner, imaging devices, hybridization machine, hybridization chamber, thermocycler, centrifuge, reaction tubes, centrifuge tubes, filter, rotator, mixer, beads, FACS, laser, particularly laser for microdissections, balloon instrument, magnetic devices for scientific purposes; diagnostic and testing apparatus and instruments, namely, syringe, needle, biopsy needle, array, particularly DNA-array, array reader, scanner, imaging devices, hybridization machine, hybridization chamber, thermocycler, centrifuge, reaction tubes, centrifuge tubes, filter, rotator, mixer, beads, FACS, laser, particularly laser for microdissections, balloon instrument, magnetic devices for the development of medicaments, for the identification of expressed genes, particularly abnormal expressed genes; Diagnostic and testing apparatus and instruments for the analysis of the gene expression patterns of single cells and cell culture lines, mammalian cells and human cells, of cells of microscopic small biopsies of animal and human tissues, of cells out of tissue samples prepared with a laser, of cells isolated with FACS or with receptor/ligand binding, of chemical fixed tissue, of micrometastatic cells, of bone marrow cells, of samples from body fluids, from amniotic fluid, liquor, blood, lymph, urine or bowel movements, of stem cells, cloned single cells or differentiated stages from specific cells
- GS0101: Medical diagnostic instruments, namely, syringe, needle, biopsy needle, array, particularly DNA-array, array reader, scanner, Imaging devices, hybridization machine, hybridization chamber, thermocycler, centrifuge, reaction tubes, centrifuge tubes, filter, rotator, mixer, beads, FACS, laser, particularly laser for microdissections, balloon instrument, magnetic devices for diagnosing tumors, coronary heart diseases, particularly restenosis, minimal residual diseases, infectious diseases, autoimmune diseases; diagnostic and testing apparatus for medical purposes, namely, syringe, needle, biopsy needle, array, particularly DNA-array, array reader, scanner, imaging devices, hybridization machine, hybridization chamber, thermocycler, centrifuge, reaction tubes, centrifuge, tubes, filter, rotator, mixer, beads, FACS, laser, particularly laser for microdissections, balloon instrument, magnetic devices, for testing for eukaryotic cells, cells of microscopic small biopsies, of cells out of tissue samples prepared with a laser, of cells isolated with FACS or with receptor/ligand binding, of chemical fixed tissue, of micrometastatic cells, of bone marrow cells, of samples from body fluids from amniotic fluid, liquor, blood, lymph, urine or bowel movement, of stem cells, cloned single cells or differentiated stages from specific cells of human tissues; medical diagnostic and testing apparatus, namely syringe, needle, biopsy-needle, array, particularly DNA-array, array reader, scanner, imaging devices, hybridization machine, hybridization chamber, thermocycler, centrifuge, reaction tubes, centrifuge tubes, filter, rotator, mixer, bead, FACS, laser, particularly laser for microdissections, balloon instrument, magnetic devices for testing animal cells for veterinary use
- GS0421: Medical services; scientific and industrial research in the field of molecular biology, bio-technology, genetics, medicine and pharmacology; medical services, namely, diagnosis of pathogenesis of animal and human diseases, cell differentiation of stem cells and other progenitor cells, steps in the development from micrometastatic cells to metastatic cells, characterization of the differentiated stages from specific cells; identification of target structures for the development of medicaments; medical services for the determination of the influence of various substances on the cell growth, proliferation, differentiation and secretion of molecules in various cell types, characterization of the differentiation of nerve cells; characterization of intracellular signal transduction pathways; determination of the influence of the co-culture of cell lines on the cell growth, proliferation, differentiation and secretion of molecules in various cell types and on differential gene expression; medical services and veterinary medicine; Development of pharmaceuticals; laboratory research in the field of identification of expressed genes, namely, abnormal expressed genes, analysis of the gene expression pattern, single cells and cell culture lines in human and mammalian cells; laboratory research in the field of cells of microscopic small biopsies of animal and human tissues, cells out of tissue samples prepared with a laser, of cells isolated with FACS or with receptor/ligand binding, of chemical fixed tissue, of micrometastatic cells, of bone marrow cells, of samples from body fluids from amniotic fluid, liquor, blood, lymph, urine or bowel movement, of stem cells, cloned single cells, differentiated stages from specific cells; Development of new technology for others in the field of scientific and industrial research
Case File Event Statements
-
11/23/2012 - 13 years ago
32 - CANCELLED SEC. 8 (6-YR)
Type: C8..
-
4/18/2006 - 19 years ago
31 - REGISTERED-PRINCIPAL REGISTER
Type: R.PR
-
2/14/2006 - 20 years ago
30 - 1(B) BASIS DELETED; PROCEED TO REGISTRATION
Type: DP1B
-
1/27/2006 - 20 years ago
29 - NOTICE OF ALLOWANCE CANCELLED
Type: IUCN
-
1/9/2006 - 20 years ago
28 - PAPER RECEIVED
Type: MAIL
-
11/16/2005 - 20 years ago
27 - FAX RECEIVED
Type: FAXX
-
9/7/2005 - 20 years ago
26 - FAX RECEIVED
Type: FAXX
-
8/11/2005 - 20 years ago
25 - FAX RECEIVED
Type: FAXX
-
8/3/2005 - 20 years ago
24 - EXTENSION 2 GRANTED
Type: EX2G
-
7/22/2005 - 20 years ago
23 - EXTENSION 2 FILED
Type: EXT2
-
7/22/2005 - 20 years ago
22 - TEAS EXTENSION RECEIVED
Type: EEXT
-
1/18/2005 - 21 years ago
21 - EXTENSION 1 GRANTED
Type: EX1G
-
1/18/2005 - 21 years ago
20 - EXTENSION 1 FILED
Type: EXT1
-
1/18/2005 - 21 years ago
19 - TEAS EXTENSION RECEIVED
Type: EEXT
-
1/10/2005 - 21 years ago
18 - PAPER RECEIVED
Type: MAIL
-
7/27/2004 - 21 years ago
17 - NOA MAILED - SOU REQUIRED FROM APPLICANT
Type: NOAM
-
5/4/2004 - 21 years ago
16 - PUBLISHED FOR OPPOSITION
Type: PUBO
-
4/14/2004 - 21 years ago
15 - NOTICE OF PUBLICATION
Type: NPUB
-
2/11/2004 - 22 years ago
14 - APPROVED FOR PUB - PRINCIPAL REGISTER
Type: CNSA
-
11/17/2003 - 22 years ago
13 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA
-
11/20/2003 - 22 years ago
12 - PAPER RECEIVED
Type: MAIL
-
11/5/2003 - 22 years ago
11 - CASE FILE IN TICRS
Type: CFIT
-
5/22/2003 - 22 years ago
10 - FINAL REFUSAL MAILED
Type: CNFR
-
3/17/2003 - 23 years ago
9 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA
-
3/17/2003 - 23 years ago
8 - PAPER RECEIVED
Type: MAIL
-
1/3/2003 - 23 years ago
7 - LETTER OF SUSPENSION MAILED
Type: CNSL
-
10/22/2002 - 23 years ago
6 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA
-
10/25/2002 - 23 years ago
5 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA
-
10/25/2002 - 23 years ago
4 - PAPER RECEIVED
Type: MAIL
-
4/24/2002 - 23 years ago
3 - NON-FINAL ACTION MAILED
Type: CNRT
-
4/17/2002 - 23 years ago
2 - ASSIGNED TO EXAMINER
Type: DOCK
-
1/22/2002 - 24 years ago
1 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA