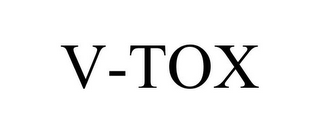Information
-
 Trademark
Trademark
-
 87293856
87293856
-
Serial Number
87293856
-
Registration Number
5483628
-
International Classifications
- 9 - Scientific, nautical, surveying, photographic, cinematographic, optical, weighing, measuring, signalling, checking (supervision), life-saving and teaching apparatus and instruments
- 34 - Tobacco
- 35 - Advertising
- 37 - Building construction
- 40 - Treatment of materials
- 42 - Scientific and technological services and research and design relating thereto
- 45 - Legal services
-
Filing Date
January 09, 2017
8 years ago
-
Registration Date
June 05, 2018
6 years ago
-
Transaction Date
January 14, 2025
4 months ago
-
Status Date
December 20, 2024
5 months ago
-
Published for Opposition Date
November 28, 2017
7 years ago
-
Location Date
June 05, 2018
6 years ago
-
Status Code
710
-
Current Location
PUBLICATION AND ISSUE SECTION
Employee Name
FERRELL, STEVEN
-
Attorney Docket Number
174558
Attorney Name
Holly M. Ford Lewis
Law Office Assigned Location Code
N30
-
Owners
Mark Drawing Code
4
Mark Identification
V-TOX
Case File Statements
- GS0401: Treatment of materials, namely, treatment of materials through extraction and purification processes in the nature of extraction of chemical substances from plant matter, purification and extraction of nicotine from plant matter, treatment of plant extracts for use in electronic smoking devices, and anaerobic processing of plant extracts; manufacturing services for others in the field of electronic smoking devices and parts, fittings, and accessories therefor; contract manufacturing services in the field of electronic smoking devices and parts, fittings, and accessories therefor; contract manufacturing services, namely, the manufacture of bespoke formulations in the nature of liquids and flavorings for electronic smoking devices; contract manufacturing of electronic smoking devices, ampoules, cartridges and refill cartridges for electronic smoking devices and parts, fittings and accessories therefor; manufacture of formulations for use in electronic smoking devices; manufacture of nicotine-containing solutions and nicotine-free solutions for electronic smoking devices; manufacturing of product variations, namely, manufacturing of formulations for use in electronic smoking devices having different compositions; providing technical advice related to the manufacture of medicinal products, medical devices, and electronic smoking devices
- GS0091: Parts, components and electronic apparatus for electronic smoking devices in the nature of batteries and chargers for electronic cigarettes
- GS0371: Providing technical advice related to the repair of medicinal products, medical devices, and electronic smoking devices
- GS0351: Business strategic planning services in the medicinal products, medical devices, and electronic smoking devices industry; regulatory submission management, namely, assisting others in preparing and filing applications for medical devices with governmental regulatory bodies in the nature of assessment and investigation of assets, preparation and review of reports and assessments for regulatory purposes; regulatory submission management, namely, assisting others in preparing and filing applications and renewals for product licenses with governmental regulatory bodies; regulatory submission management, namely, assisting others in preparing and filing applications for approval of formulations in the nature of liquids and flavorings for use in electronic smoking devices with governmental regulatory bodies
- GS0341: Tobacco; smokers' articles, namely, electronic cigarette holders; matches; electronic cigarettes; tobacco products, namely, smoking tobacco; tobacco substitutes; cigarettes containing tobacco substitutes, not for medical purposes; electronic smokers' article, namely, smokers' mouthpieces for electronic cigarettes; electronic cigars; electronic smokers' pipes; electronic smoking devices, namely, electronic smoking pipes; refill liquid and other fillers for electronic smoking devices, namely, electronic cigarette liquid (e-liquid) comprised of flavorings in liquid form, other than essential oils, used to refill electronic cigarette cartridges and other flavorings, other than essentials oils, for use in electronic cigarettes; smokers' oral vaporizer ampoules, cartridges, and refill cartridges for electronic smoking devices sold empty; flavourings, other than essential oils, for use in electronic cigarettes; flavours and flavour essences, other than essential oils, for electronic cigarettes and other electronic smoking devices in the nature of cigars and pipes; personal oral vaporisers for smokers, and electronic smoking devices in the nature of cigarettes, cigars, and pipes, and flavourings, other than essential oils and liquid nicotine solutions therefor; atomisers for electronic smoking devices in the nature of cigarettes, cigars, and pipes and cartomisers, namely, combination electronic cigarette refill cartridges sold empty and atomizers, sold as a component of electronic cigarettes; liquid nicotine solutions for use in electronic smoking devices in the nature of cigarettes, cigars, and pipes; liquid nicotine-substitute solutions for use in electronic smoking devices, namely, electronic cigarette (e-liquid) comprised of propylene glycol and vegetable glycerin; nicotine-free solutions for use in electronic smoking devices, namely, electronic cigarette (e-liquid) comprised of propylene glycol and vegetable glycerin; electronic cigarette cases; parts, components and electronic apparatus for electronic smoking devices in the nature of refill cartridges sold empty; replacement parts, fittings, and accessories for all of the aforementioned
- GS0451: Legal services; physical security guard services for the protection of tangible property and individuals; safety evaluation, namely, security threat analysis for protecting personal safety; legal services relating to medical products, medical devices and electronic smoking devices; advisory services relating to regulatory compliance in the field of medical products, medical devices, and electronic smoking devices; monitoring and tracking of regulatory requirements in non-clinical and clinical pharmaceutical and medical device trials for regulatory compliance purposes; regulatory compliance consulting in the field of medical products, medical devices, and electronic smoking devices in the nature of assessment and investigation of assets, preparation and review of reports and assessments for regulatory purposes; providing information and advice regarding regulatory compliance in the field of medical products, medical devices, and electronic smoking devices
- GS0421: Scientific and technological services, namely, scientific research, analysis, and testing in the field of electronic smoking devices and parts, fittings, and accessories therefor; industrial analysis and research services, namely, analysis of the goods of others to assure compliance with industry standards and industrial research in the field of electronic smoking devices and parts, fittings, and accessories therefor; design and development of computer hardware and software; research, consultancy and advisory services in relation to medical devices; product safety testing; information services relating to the safety of chemicals used in electronic smoking devices; technical advice relating to the operation of medicinal products, medical devices, and electronic smoking devices; research of pharmaceuticals; pharmaceutical product development, evaluation and testing; product development for others; safety testing of products; testing of electronic smoking device chemicals; research relating to chemicals for use in electronic smoking articles; chemical research and analysis; laboratory analysis in the field of electronic smoking devices and parts, fittings, and accessories therefor; laboratory research in the field of electronic smoking devices and parts, fittings, and accessories therefor; scientific laboratory services; research relating to electronic smoking devices and parts, fittings and accessories therefor; product design and development in the nature of formulation of formulations for use in electronic smoking devices; product design and development in the nature of formulation of nicotine-containing solutions and nicotine-free solutions for electronic smoking devices
Case File Event Statements
-
1/12/2017 - 8 years ago
1 - NEW APPLICATION ENTERED
Type: NWAP
-
1/17/2017 - 8 years ago
2 - NEW APPLICATION OFFICE SUPPLIED DATA ENTERED
Type: NWOS
-
4/1/2017 - 8 years ago
3 - ASSIGNED TO EXAMINER
Type: DOCK
-
10/5/2017 - 7 years ago
9 - TEAS/EMAIL CORRESPONDENCE ENTERED
Type: TEME
-
4/5/2017 - 8 years ago
4 - PRIORITY ACTION WRITTEN
Type: CPRA
-
4/5/2017 - 8 years ago
6 - NOTIFICATION OF PRIORITY ACTION E-MAILED
Type: GPRN
-
4/5/2017 - 8 years ago
5 - PRIORITY ACTION E-MAILED
Type: GPRA
-
10/4/2017 - 7 years ago
7 - TEAS RESPONSE TO OFFICE ACTION RECEIVED
Type: TROA
-
10/4/2017 - 7 years ago
8 - CORRESPONDENCE RECEIVED IN LAW OFFICE
Type: CRFA
-
10/6/2017 - 7 years ago
10 - EXAMINERS AMENDMENT -WRITTEN
Type: CNEA
-
10/6/2017 - 7 years ago
13 - EXAMINER'S AMENDMENT ENTERED
Type: XAEC
-
10/6/2017 - 7 years ago
12 - NOTIFICATION OF EXAMINERS AMENDMENT E-MAILED
Type: GNEN
-
10/6/2017 - 7 years ago
11 - EXAMINERS AMENDMENT E-MAILED
Type: GNEA
-
10/6/2017 - 7 years ago
14 - APPROVED FOR PUB - PRINCIPAL REGISTER
Type: CNSA
-
10/20/2017 - 7 years ago
15 - ASSIGNED TO LIE
Type: ALIE
-
10/26/2017 - 7 years ago
16 - LAW OFFICE PUBLICATION REVIEW COMPLETED
Type: PREV
-
11/8/2017 - 7 years ago
17 - NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED
Type: NONP
-
11/28/2017 - 7 years ago
18 - PUBLISHED FOR OPPOSITION
Type: PUBO
-
11/28/2017 - 7 years ago
19 - OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED
Type: NPUB
-
1/23/2018 - 7 years ago
20 - NOA E-MAILED - SOU REQUIRED FROM APPLICANT
Type: NOAM
-
3/28/2018 - 7 years ago
21 - TEAS DELETE 1(B) BASIS RECEIVED
Type: D1BR
-
3/28/2018 - 7 years ago
23 - NOTICE OF ALLOWANCE CANCELLED
Type: IUCN
-
4/27/2018 - 7 years ago
22 - CASE ASSIGNED TO INTENT TO USE PARALEGAL
Type: AITU
-
4/27/2018 - 7 years ago
24 - 1(B) BASIS DELETED; PROCEED TO REGISTRATION
Type: DP1B
-
6/5/2018 - 6 years ago
25 - REGISTERED-PRINCIPAL REGISTER
Type: R.PR
-
6/5/2023 - a year ago
26 - COURTESY REMINDER - SEC. 8 (6-YR) E-MAILED
Type: REM1
-
12/20/2024 - 5 months ago
27 - CANCELLED SEC. 8 (6-YR)
Type: C8..